

How To Improve Our Tendon Health with Training
We chat with Dr. Keith Baar about the delicate balance between tendon stiffness and laxity.
November 3, 2023

Keith Baar, PhD
Dr. Keith Baar is a Professor at the University of California Davis and renowned tendon training researcher and expert. Dr. Baar currently heads the Functional Molecular Biology Laboratory (FMBLab). His work is focused on how loading and nutrition alter connective tissue health and performance. Over the last 15 years, Dr. Baar has worked with elite athletes as a scientific advisor to the Chelsea Football Club, USA Track and Field, Paris Saint-Germain Football Club, British Cycling, and more.
It’s not something you see every day — a robotic fish powered using muscles from frog legs. Dr. Keith Baar’s journey into the realm of tendon research began as a postdoctoral researcher in the lab of Bob Dennis, which was at the cutting edge of robotics and biology. When the frog’s muscles twitched, the fishbot’s tail swayed and propelled it forward. But its swim was short-lived.1
“The muscles would always fail at the muscle-machine interface,” said Dr. Baar, reflecting on those early days. Their team needed a better design to connect the muscles to the hard mechanical surface.
The team drew their inspiration from our own biological architecture — tendons, which connect muscles and bones. They transmit force from muscles to bones and store elastic energy to improve the efficiency of movement. Ligaments, on the other hand, attach bone to bone and provide structural support.
Yet tendons exhibit a remarkable duality; they are stiffer where they meet bone and stretchier where they connect to muscle.
“What the tendon is doing is providing a transition,”2 said Dr. Baar. “It’s transitioning the mechanics of the muscle to the mechanics of the bone — going from something soft and stretchy to something hard. And that’s what we were missing in our little robots, and that’s why our muscles always pulled off the robot.”
The fishbot jump-started Dr. Baar’s exploration into tendon and ligament (connective tissue) research. He learned that a significant amount of time away from sports is due to musculoskeletal injuries. And this made him curious: “What can we do with our exercise and nutrition to maintain healthy tendons? Can we optimize tendons so that athletes can perform at their best?”
How Do Tendon Injuries Happen?
In athletics and performance research, you’ll often hear a term thrown around — “jerk.” It’s not just an insult flung at the person you don’t like, but a type of movement that is sharp and sudden. In physics and engineering, jerk is defined as the rate of change of acceleration.
Dr. Baar provides the example of playing tennis on the first beautiful day of the year. To hit a ball, the player’s racket is accelerating in one direction while the ball is careening through the air in the opposite direction. When the racket smashes the ball, you get high jerk (a rapid change in acceleration). Without proper warm-up and training, the player is potentially at risk of elbow pain.
“If they don’t ramp up into the action (by slowly increasing the amount or intensity of training over days to weeks), it could give them an overuse tendon injury,” said Dr. Baar.

Another sport than can produce high jerk is basketball. “If I’m a basketball player and I go for a rebound. Bang, I’m going to land. Then I’m going to jump to put it back up. That quick acceleration in two directions, that’s your jerk,” said Dr. Baar. Credit: Adobe Photos
One reason for this is because consistent high jerk, such as found in fast exercises, increases tendon stiffness. This in turn ups an athlete’s force production, meaning their movements will become more powerful. However, a delicate equilibrium exists between tendon stiffness and laxity. Stiffer tendons produce better strength, power, and speed. And yet, when tendon stiffness surpasses the muscle’s ability to resist, an athlete is at a higher chance of pulling their muscle.
“In general, what you want for your ligaments is for them to be as stiff and strong as possible,” said Dr. Baar. “The stiffer the ligament the better the structural support. The same is not true of tendons. If tendons are too stiff, muscle injuries are more likely to occur.”3,4

Training Tip
If an athlete has a history of non-contact muscle pulls and you want to decrease tendon stiffness, consider incorporating slower exercises such as yoga, slow and heavy weight training, or isometric holds into their training plan while reducing the frequency of fast, powerful movements.
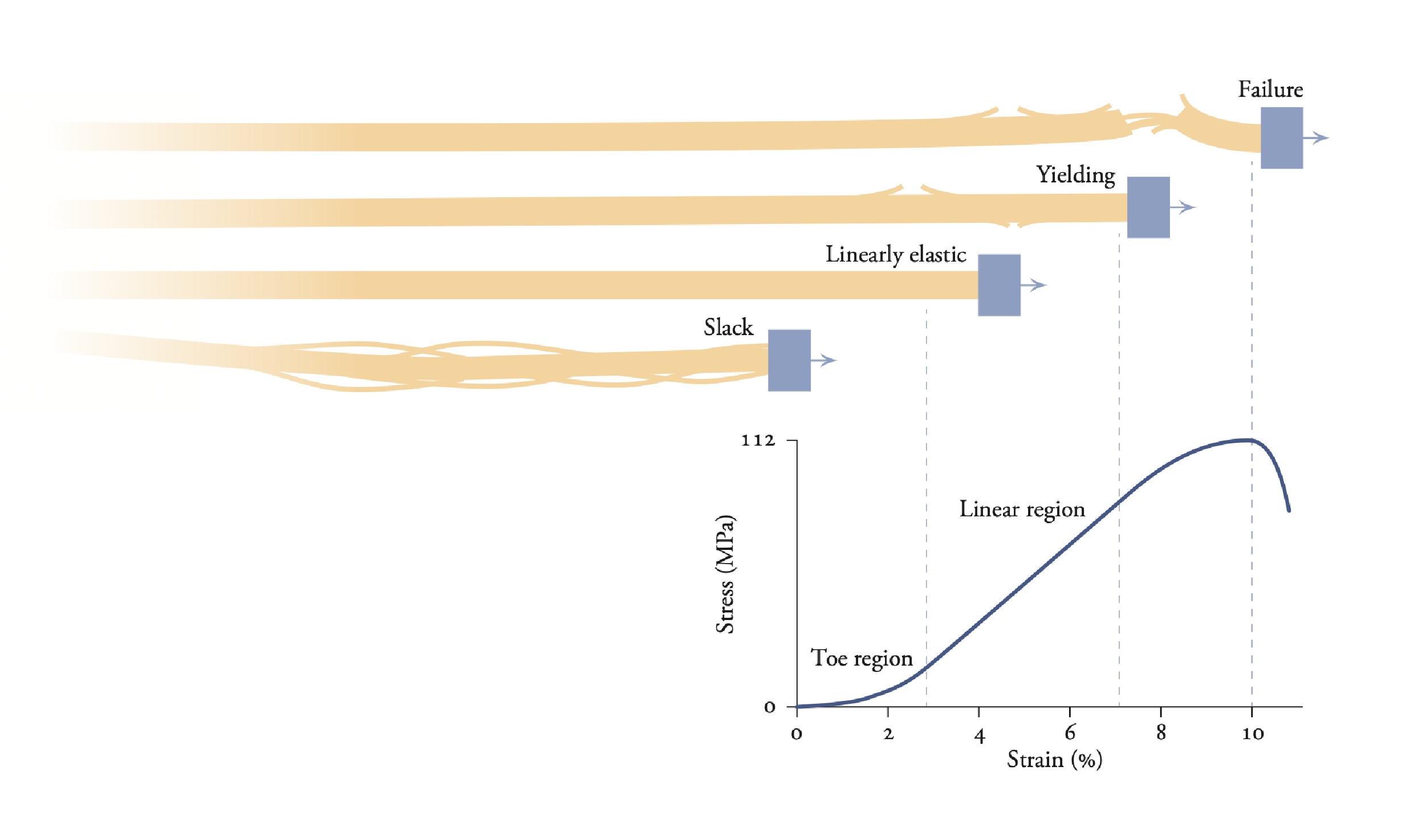
This graph shows how the behavior of tendon changes under different amounts of stress, or external forces applied to a given cross-sectional area. At lower stresses, there is less strain, or fraction of length change, and the tendon is slack or behaves like an elastic band. Above approximately 10% change in tendon length, the tendon experiences “failure.” David Delp | Biomechanics of Movement
“Connective tissue stiffness is extremely important for everyone, whether you are an athlete or just training for life,” said Dr. Baar. Stiffness influences both our performance and the likelihood we get injured. Nowhere is this more painfully obvious than between male and female athletes.
“What we see in male athletes is that their tendons and ligaments are, on average, stiffer than female athletes and as a result they suffer more muscle pulls,” said Dr. Baar. “Women, on the other hand, suffer 4- to 8-times more anterior cruciate ligament (ACL) ruptures than men. Hormones are one of the many reasons why. In general, male hormones increase stiffness, whereas female hormones decrease stiffness.”5
Hormonal fluctuations during a woman’s menstrual cycle can affect the laxity of her ligaments. This laxity is determined by the balance of her hormones estrogen, progesterone and relaxin. In contrast, men tend to pull more muscles because testosterone can increase connective tissue stiffness so it is more common for their tendons to become stiffer than their muscle is strong.6
With @IAAFDoha2019 one week a away, I figured it might be interesting to look at why there might be hamstring pulls in the men’s 200 and 400m sprints. Just a simple demonstration as to why increases in tendon stiffness (for the same muscle strength) cause muscle pulls/ruptures. pic.twitter.com/ITuMvu7V1X
— Keith Baar (@MuscleScience) September 20, 2019
How to Exercise Tendons and Ligaments
For years, tendons and ligaments were considered passive structures that didn’t respond to exercise. Over the years, a different truth emerged. A tendon’s outer layers showcase a remarkable ability to adapt with exercise. What does not change as significantly in adulthood is the collagen core, although more research is needed to confirm this is true across all tendons in the body.7,8,9
To explore what parameters affect the outside of the tendon, Dr. Baar’s laboratory and others engineered artificial ligaments from human anterior cruciate ligament cells and started to exercise them. These experiments involved stretching the connective tissue at different frequencies, intensities, and durations to determine how the molecular response that stimulates collagen production is affected by exercise. Surprisingly, the frequency and intensity of loading didn’t significantly affect the molecular response. Instead, the key factor was time.10
After 10 minutes of loading, the molecular response peaked. If loading continued, the molecular signals began to switch off. Dr. Baar’s team showed that it took around 6 hours for the cells to become responsive to exercise again.
Based on these findings, Dr. Baar developed a loading routine that involves 6-10 minutes of activity followed by at least a 6-hour period of rest. Over a span of 5 days, the engineered ligaments subjected to this protocol produced higher levels of collagen compared to those that were exercised continuously.11

Dr. Baar’s Advice
Dr. Baar’s research suggests short periods of activity (<10 minutes) to a tendon/ligament, regardless of the load, will activate the cells in connective tissue. When combined with periods of rest that are >8 hours, optimum connective tissue health is possible.12,13
Note: Dr. Baar’s team has used this protocol for several case studies, but there are no large clinical trial results to draw on at this time.
Why does this work? When you exercise you increase the production of collagen and many other proteins important for connective tissue adaptation. However, the longer you exercise the more mechanical damage can occur to these tissues. To optimize adaptations, the goal is to provide the maximal signal to the cells at the minimal amount of mechanical damage. This is where the short exercise bouts can be useful for connective tissues like tendons, ligaments, and bones.14
Another way that our exercise affects connective tissue stiffness is by regulating the crosslinks between collagen molecules. Since crosslinks restrict the sliding of individual collagen fibrils alongside one another, having more crosslinks results in stiffer connective tissues. Within a tendon, there are more crosslinks near the bone (the stiffer portion) than the muscle, helping to create the mechanical transition Dr. Baar discussed earlier.
Exercise regulates crosslinking by physically breaking these attachments and then rebuilding them using an enzyme known as lysyl oxidase. When we exercise at fast speeds, we don’t break as many crosslinks as when we move slowly against a heavy weight. Both types of exercise increase lysyl oxidase in the tendon.15 Therefore, with fast movements we keep the crosslinks we have and make more, whereas with heavy/slow exercises we break crosslinks and then make new ones.
If the goal is greater performance, fast movements increase crosslinks near the muscle. Since the matrix is stiffer it doesn’t stretch as much from the same load. The less a tissue stretches at a given load, the faster it transmits force and the faster an athlete can run/jump/throw. The only drawback is that it can make the attached muscle more prone to injury.
A person’s athletic history also plays a role in how they need to train to maintain tendon health. If an athlete has a history of muscle pulls, then they may need to incorporate more slow, heavy weights and fewer of the fast, light movements. On the other hand, if an athlete is in the off-season, where they are not getting the plyometric load of playing their sport, then gradually adding those fast moves before the season starts is key to building power.
“You’re likely never going to be a great golfer if all you do is slow, heavy loads on your elbow,” said Dr. Baar. “You may never have golfer’s elbow, but you’re also not going to get as much power through your swing as someone who practices fast movements. What you want to do is combine the two types of training.”
Ultimately, an athlete’s tendon journey needs to be a personalized plan based on their genetics, medical history (i.e., injuries), the sport they play, and where they are in the season.
Hear more from Dr. Keith Baar on his latest research on tendon development and repair

Flash Q&A with Keith Baar
What is your favorite athletic activity?
I love playing basketball, but because it is hard to find games I end up running and lifting weights to stay fit.
Misconception about tendons you would like to debunk?
That you can’t fix them. We know that we can fix tendons completely. The key is to continue to put the right loads through the tissue even after the pain diminishes.
Get Deeply Researched Insights on Human Performance
Join our mailing list to get actionable performance tips and nuanced explanations of the science.
Citations
- Herr, H., & Dennis, R. (2004). A swimming robot actuated by living muscle tissue. Journal of Neuroengineering and Rehabilitation, 1(6). https//doi.org/10.1186/1743-0003-1-6.
- Arruda, E. M., Calve, S., Dennis, R. G., Mundy, K., & Baar, K. (2006). Regional variation of tibialis anterior tendon mechanics is lost following denervation. Journal of Applied Physiology (1985), 101(4), 1113-1117. https//doi.org/10.1152/japplphysiol.00612.2005.

Does this research apply to humans?
Since the study conducted by Arruda, et al. used tibialis anterior tendons from rats, the findings may not accurately reflect what occurs in all human tendons. What the study does do is provide preliminary evidence for specific tendon mechanics, adding to observations made in other studies, such as in the rat Achilles tendon.
- LaCroix, A. S., Duenwald-Kuehl, S. E., Lakes, R. S., & Vanderby, R. Jr. (2013). Relationship between tendon stiffness and failure: a metaanalysis. Journal of Applied Physiology (1985), 115(1), 43-51. https//doi.org/10.1152/japplphysiol.01449.2012.
- Myer, G. D., et al. (2008). The effects of generalized joint laxity on risk of anterior cruciate ligament injury in young female athletes. The American Journal of Sports Medicine, 36(6), 1073-1080. https//doi.org/10.1177/0363546507313572.
- Chidi-Ogbolu, N., & Baar, K. (2019). Effect of estrogen on musculoskeletal performance and injury risk. Frontiers in Physiology, 9, 1834. https//doi.org/10.3389/fphys.2018.01834
- Raj, R. D., Fontalis, A., Grandhi, T. S. P., Kim, W. J., Gabr, A., & Haddad, F. S. (2023). The impact of the menstrual cycle on orthopaedic sports injuries in female athletes. Bone & Joint Journal, 105-B(7), 723-728. https//doi.org/10.1302/0301-620X.105B7.BJJ-2022-1262.R2.
- Heinemeier, K. M., Schjerling, P., Heinemeier, J., Magnusson, S. P., & Kjaer, M. (2013). Lack of tissue renewal in human adult Achilles tendon is revealed by nuclear bomb (14)C. The FASEB Journal, 27(5), 2074-2079. https//doi.org/10.1096/fj.12-225599.
- Grinstein, M., Dingwall, H. L., O’Connor, L. D., Zou, K., Capellini, T. D., & Galloway, J. L. (2019). A distinct transition from cell growth to physiological homeostasis in the tendon. eLife, 8, e48689. https//doi.org/10.7554/eLife.48689.
- Zhang, C., Svensson, R. B., Couppé, C., Schjerling, P., Skovgaard, D., Kjaer, M., & Magnusson, S. P. (2020). Regional differences in turnover, composition, and mechanics of the porcine flexor tendon. Connective Tissue Research, 61(5), 475-484. https//doi.org/10.1080/03008207.2019.1620222.
- Baar, K. (2017). Minimizing injury and maximizing return to play: lessons from engineered ligaments. Sports Medicine, 47(Suppl 1), 5-11. https//doi.org/10.1007/s40279-017-0719-x.
- Paxton, J. Z., Hagerty, P., Andrick, J. J., & Baar, K. (2012). Optimizing an intermittent stretch paradigm using ERK1/2 phosphorylation results in increased collagen synthesis in engineered ligaments. Tissue Engineering Part A, 18(3-4), 277-284. https//doi.org/10.1089/ten.TEA.2011.0336.

How similar are engineered ligaments to human tendons?
It’s important to note that engineered ligaments, as used in this study by Paxton, et al., are a wonderful model, but they are not perfect. They are much more similar to embryonic tendons (lots of cells and a small weak matrix) than adult tendons.
- Power, D., Haddad, F., Wallis, S., & Baar, K. (2023). Ramping isometrics for accelerated return to play following hamstring tendon repair: a case study. Journal of Elite Sport Performance. Published online June 1, 2023. https//doi.org/10.54080/MOMV6327.
- Baar, K. (2019). Stress relaxation and targeted nutrition to treat patellar tendinopathy. International Journal of Sport Nutrition and Exercise Metabolism, 1-5. https//doi.org/10.1123/ijsnem.2018-0231
- Min, S. K., Oh, T., Kim, S. H., Cho, J., Chung, H. Y., Park, D. H., & Kim, C. S. (2019). Position statement: exercise guidelines to increase peak bone mass in adolescents. Journal of Bone Metabolism, 26(4), 225-239. https//doi.org/10.11005/jbm.2019.26.4.225.
- Heinemeier, K. M., Olesen, J. L., Haddad, F., Langberg, H., Kjaer, M., Baldwin, K. M., & Schjerling, P. (2007). Expression of collagen and related growth factors in rat tendon and skeletal muscle in response to specific contraction types. The Journal of Physiology, 582(Pt 3), 1303-1316. https//doi.org/10.1113/jphysiol.2007.127639.
Playbook Terms of Use & Copyright
©2023-2024



